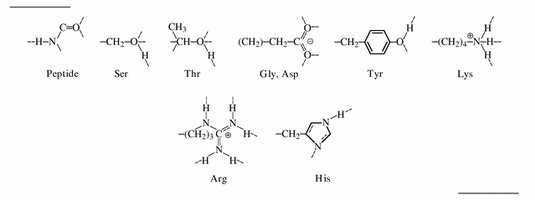
Bonding is a phenomenon that occurs in many aspects of chemistry. Because silk is a polypeptide, it contains many amino acids and varying single elements within these acids. These single elements are responsible for bonding and holding the molecules together. There are three main types of bonds present in silk (Sashina, Bochek, Kirichenko, 2006).
HYDROGEN BONDS
Hydrogen bonds are the strongest type of intermolecular bonds. They occur when hydrogen is present, along with nitrogen, oxygen and/or fluorine. In Figure 4, every amino acids contains hydrogen, proving it is the most important element in bonding.
THE CARBOXYL GROUP
A carboxyl group is a functional group containing a carbon double bonded to an oxygen. There is a carboxyl group in Figure 4 on the peptide molecule. This bond is highly polar, resulting in dipole attractions (Sashina, Bochek, Kirichenko, 2006).
INTERMOLECULAR FORCES
These forces are also known as intermolecular forces and are quite weak compared to hydrogen bonds. There are three types of intermolecular forces.
- London Dispersion
- Dipole- Dipole
- Hydrogen Bonds
London Disperion
London forces occur between two non-polar molecules. Because silk is a polypeptide, it contains many molecules including non-polar ones. These non-polar molecules attract each other and create attractive forces, which aids in holding the protein together.
Dipole-Dipole
Dipole-dipole forces are stronger than london forces, but not as strong as hydrogen bonds. These bonds occur between polar molecules. This type of bonding occurs because the positive end of one dipole is attracted to the negative end of another dipole, and vice versa. Generally, a substance does not contain both london forces and dipole-dipole forces, but because silk is a large protein and different subsections, both polar and non-polar molecules are present (Sashina, Bochek, Kirichenko, 2006).
Another factor that affects bonding in proteins is the tertiary structure. Because silk can form beta-pleated sheets and alpha helixes, it can form similar bonds on different angles. This idea is shown in Figure 3 (under Structural Levels). More bonds occur in beta-pleated sheets because they can be packed very tightly as well as fold together, while alpha helixes cannot unwind and rewind closer together.