Polypeptide Structure Development
|
Polypeptides generally have three to four different structural levels. These levels depend on the amino acids that make up the protein. The four levels include...
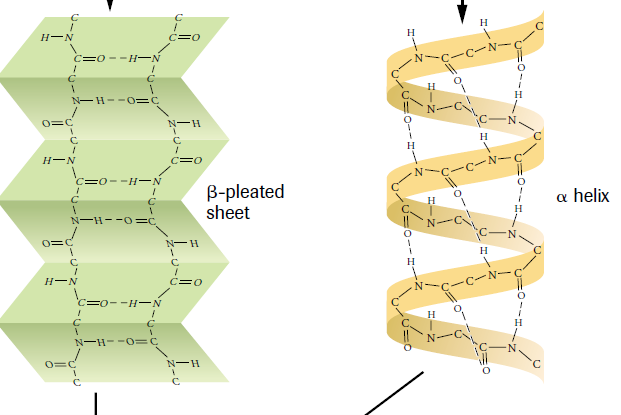
1. Primary Structure 2. Secondary Structure 3. Tertiary Structure 4. Quaternary Structure Primary Structure The primary structure is a sequence of amino acids. Fibroin and sericin would be examples of primary structure proteins. Secondary Structure In this stage, the proteins coil or bend into an alpha helix of a beta pleated sheet. Figure 3 displays the difference between these structures. Generally, the formation of beta pleated sheets occur in structural proteins, including silk cocoons. Tertiary Structure During the third stage, the protein chain folds hundreds of times to form a small ball. Hydrogen bonding is a key component to this stage. As the proteins fold on themselves, the elements move much closer together which results in very strong bonds. Quaternary Structure In order for this stage to occur, there must be three or more proteins present. These proteins then form a complex. This phase does not always occur and it does not occur in silk because there are only two proteins present in silk cocoons. |
Key
Grey -> The ideas can be related to all polypeptides Blue -> These ideas directly relate to silk |
Secondary Structures
Because fibroin is the main component of silk, it can be subdivided into three sections...
Each section is made up of different amino acids resulting in different structures (Sashina, Bochek, Kirichenko, 2006).
- Silk 1
- Silk 2
- Amorphous (Sashina, Bochek, Kirichenko, 2006)
Each section is made up of different amino acids resulting in different structures (Sashina, Bochek, Kirichenko, 2006).
Silk 1
Silk 1 is part of the crystalline area and makes up about 13% of the material (Sashina, Bochek, Kirichenko, 2006). This section has different amino acids compared to Silk 2, resulting in an alpha-helix formation (Sashina, Bochek, Kirichenko, 2006). This "spiral" results in different bonds than a beta pleated sheet formation (Sashina, Bochek, Kirichenko, 2006). This variance in bonds is displayed in Figure 3.
Silk 2
The second division of silk is known as Silk 2. It is also part of the crystalline area but it is much larger, occupying about 55% of the fiber (Sashina, Bochek, Kirichenko, 2006). This portion of silk forms pleated sheets (Sashina, Bochek, Kirichenko, 2006). Because Silk 2 forms the majority of the fiber, silk is generally known to be made up of only beta pleated sheets (Sashina, Bochek, Kirichenko, 2006).
Amorphous
The last portion of silk is known as the amorphous portion because it has no distinct shape or size (Sashina, Bochek, Kirichenko, 2006). Under a microscope, this section of silk appears as "random globules" (Sashina, Bochek, Kirichenko, 2006). The amorphous section of the silk is more commonly known as sericin (Sashina, Bochek, Kirichenko, 2006).